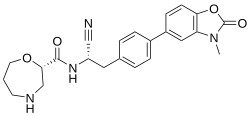
Brensocatib
 | |
| Clinical data | |
|---|---|
| Other names | AZD7986; INS1007 |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C23H24N4O4 |
| Molar mass | 420.469 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Brensocatib is an investigational new drug that is being evaluated to treat bronchiectasis.[1] It is a dipeptidyl-peptidase I (also known as cathepsin C) inhibitor.[2]
A phase 3 clinical trial, known as the ASPEN trial, was conducted to evaluate the safety and efficacy of brensocatib in patients with non-cystic fibrosis bronchiectasis.[3]
References
- ^ "Brensocatib - Insmed". AdisInsight. Springer Nature Switzerland AG.
- ^ Chalmers JD, Usansky H, Rubino CM, Teper A, Fernandez C, Zou J, et al. (October 2022). "Pharmacokinetic/Pharmacodynamic Evaluation of the Dipeptidyl Peptidase 1 Inhibitor Brensocatib for Non-cystic Fibrosis Bronchiectasis". Clinical Pharmacokinetics. 61 (10): 1457–1469. doi:10.1007/s40262-022-01147-w. PMC 9553789. PMID 35976570.
- ^ Chalmers JD, Burgel PR, Daley CL, De Soyza A, Haworth CS, Mauger D, et al. (April 2025). "Phase 3 Trial of the DPP-1 Inhibitor Brensocatib in Bronchiectasis". The New England Journal of Medicine. 392 (16): 1569–1581. doi:10.1056/NEJMoa2411664. PMID 40267423.